Fda Protocol Template
Here are some of the images for Fda Protocol Template that we found in our website database.

Free FDA Clinical Protocol Template to Edit Online

Free FDA Clinical Protocol Template to Edit Online

Fda Protocol Template

Fda Protocol Template

Fda Protocol Template

Free Test Protocol Template to Edit Online

Treatment Protocol Template Edit Online Download Example Template net

Free Performance Qualification Protocol Template to Edit Online
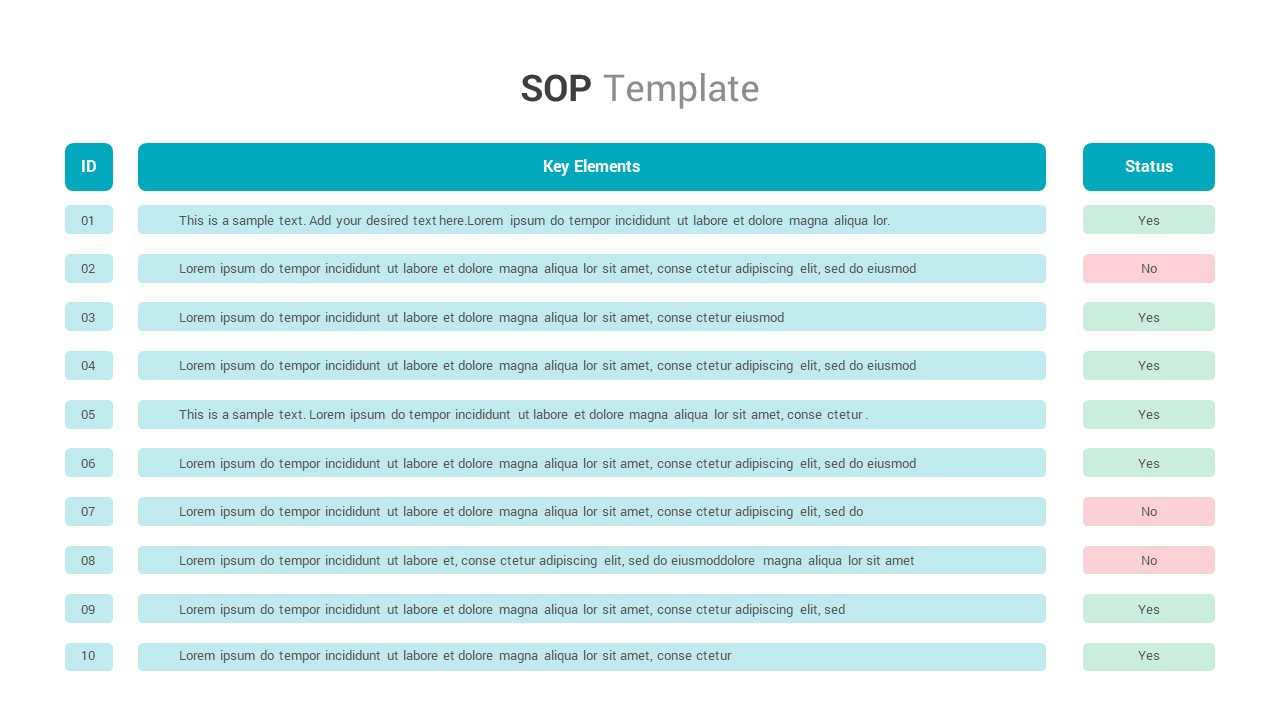
Standard Operating Protocol Template SlideBazaar

Standard Operating Protocol Template SlideBazaar

Free Study Protocol for Clinical Research Template to Edit Online
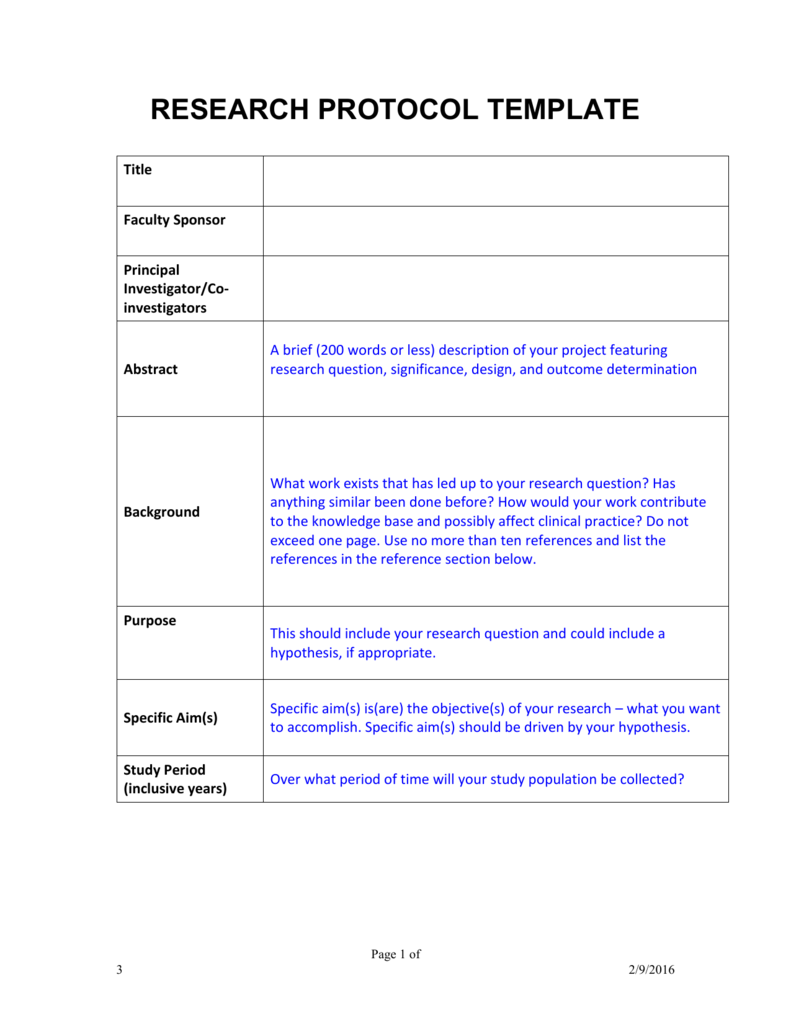
Word Protocol Template

FDA and NIH finalize clinical trial protocol template Health Law Pulse
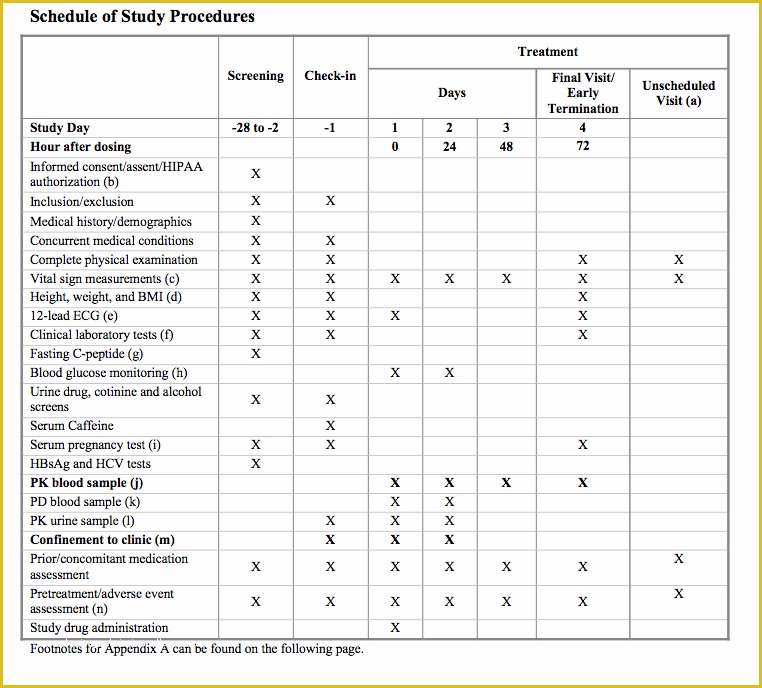
Fda Sop Template
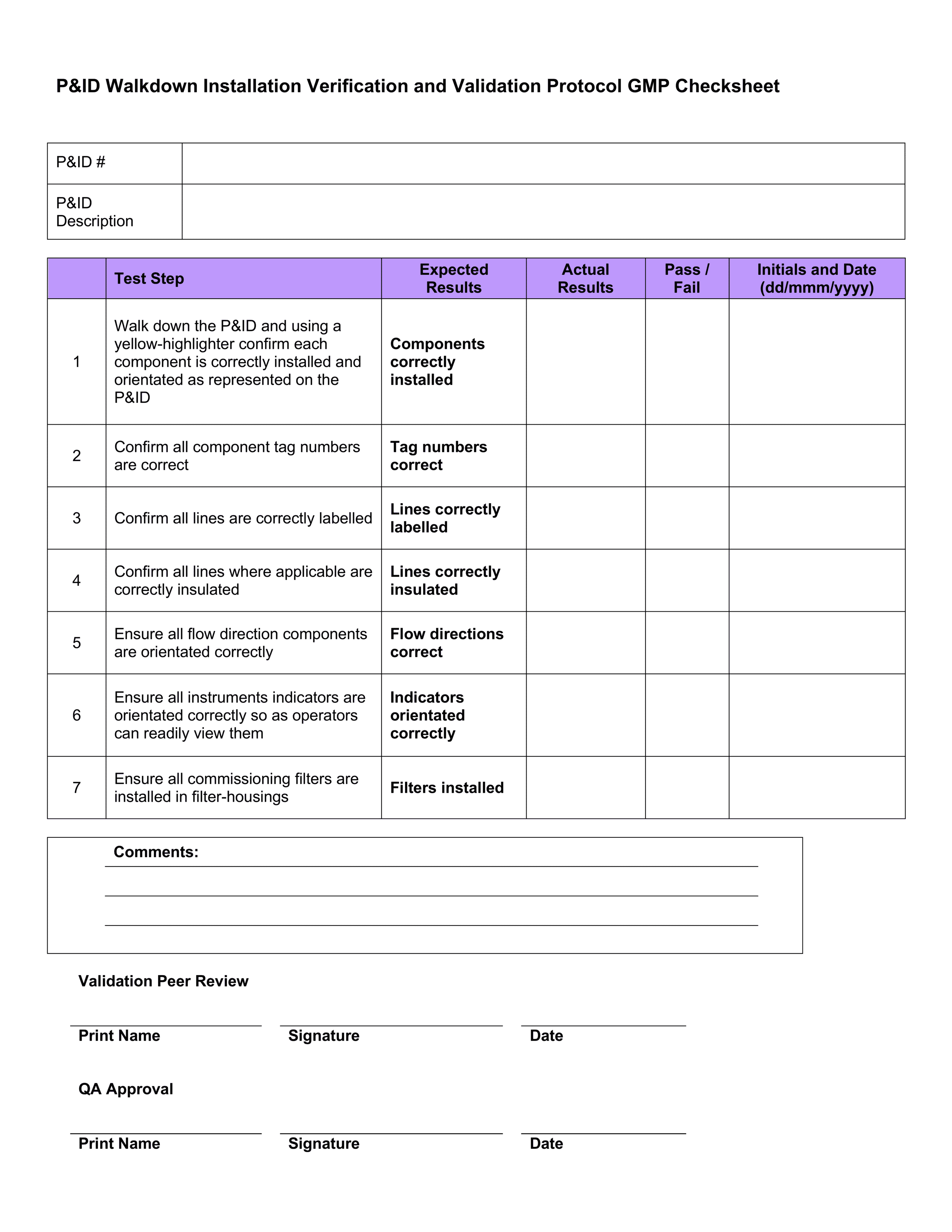
Validation Protocol Template

Fillable Online NIH FDA Phase 2 and 3 IND/IDE Clinical Trial Protocol

NIH FDA Phase 2 and 3 IND/IDE Clinical Trial Protocol Template PREFACE

NIH FDA Phase 2 and 3 Clinical Trial Protocol Template: A Course Hero

Temperature Mapping Protocol Template prntbl concejomunicipaldechinu

Fillable Online accessdata fda Order Set / Protocol Template U S

NIH FDA Protocol Template Phase 2 and 3 Clinical Trials PDF

Common Protocol Template (CPT) Initiative Collaboration with FDA and

Common Protocol Template (CPT) Initiative Collaboration with FDA and

Free Project Declaration Template to Edit Online

New FDA Protocol Deviation Guidance: Planning for the Things That Don t

Free Cleaning Service Report Template Edit Online Download

Regulation and Standard Library The FDA Group
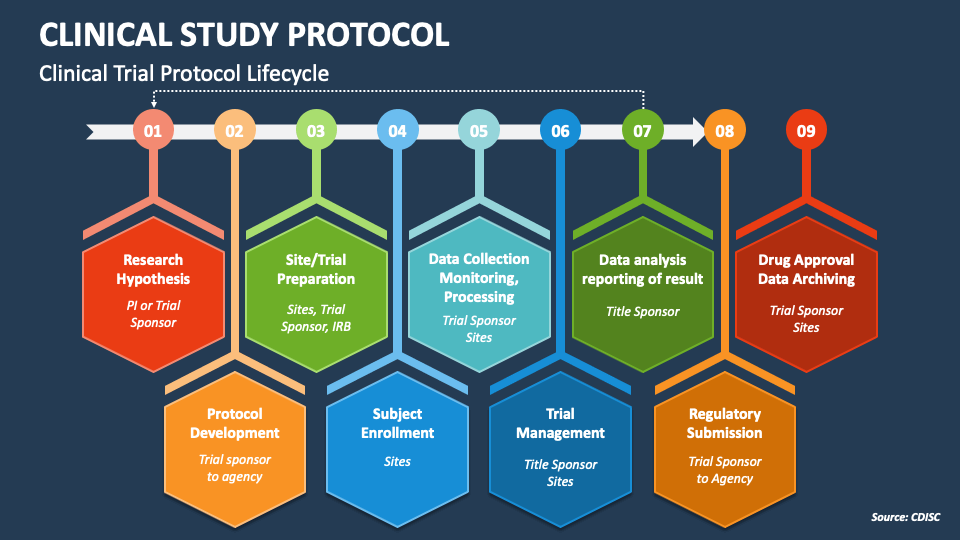
Clinical Study Protocol PowerPoint and Google Slides Template PPT Slides
Radio Advertising Template prntbl concejomunicipaldechinu gov co

Radio Advertising Template prntbl concejomunicipaldechinu gov co

PPT Collaboration with FDA and NIH PowerPoint Presentation free

PPT Collaboration with FDA and NIH PowerPoint Presentation free

Instructions for Organ Dysfunction Studies: Pharmacokinetics Course Hero

Collaboration with FDA and NIH December 2017 NIH

Collaboration with FDA and NIH Clinical Content Reuse

Fillable Online fda Warning letter Food and Drug Administration fda

Understanding the FDA s Draft Guidance on Protocol Deviations in

PSUR Submission Checklist PDF Pharmacovigilance Clinical Trial

PSUR Submission Checklist PDF Pharmacovigilance Clinical Trial

Free Clinical Trial Templates Smartsheet

Free Clinical Trial Templates Smartsheet