Eu Mdr Technical Documentation Template
Here are some of the images for Eu Mdr Technical Documentation Template that we found in our website database.

EU MDR Kit 4EasyReg

EU MDR Technical Documentation Kit 4EasyReg
EU MDR Technical Documentation requirements Kobridge

MDR Technical Documentation Thema Med

MDR Technical Documentation Thema Med

Requirements of Technical Documentation EU MDR OMC Medical

Technical Documentation for medical devices under EU MDR

Understanding Notified Body Criteria for EU MDR Technical Documentation

EU MDR Technical Documentation Checklist Celegence PDF Verification

Technical Documentation for Medical Devices Under EU MDR

Technical Documentation for Medical Devices Under EU MDR

What Notified Bodies Look for When Reviewing Your Medical Device

Provide eu mdr technical documentation of medical devices by

Provide eu mdr technical documentation of medical devices by

BUNDLE Premium Pack EU MDR Technical File Easy Medical Device School

BUNDLE Premium Pack EU MDR Technical File Easy Medical Device School

BUNDLE Premium Pack EU MDR Technical File Easy Medical Device School

BUNDLE Premium Pack EU MDR Technical File Easy Medical Device School

BUNDLE Premium Pack EU MDR Technical File Easy Medical Device School

BUNDLE Premium Pack EU MDR Technical File Easy Medical Device School

Mdr Technical File Template Printable Word Searches

Checklist for Writing Technical Documentation for EU MDR Compliance

EU MDR Consultant CE Marking Regulatory Support

Eu MDR 2017/745 Technical Documentation for CE Marking in Unnao ID

IVDR Technical Documentation

MDR assessment of Technical Documentation: Timelines TÜV SÜD

MDR 2017/745 Technical Documentation Template Checklist Easy

MDR 2017/745 Technical Documentation Template Checklist Easy

MDR 2017/745 Technical Documentation Template Checklist Easy

MDR 2017/745 Technical Documentation Template Checklist Easy

EU MDR Technical Documentation Guide Advisera

EU MDR Technical Documentation Guide Advisera

EU MDR Technical Documentation Guide Advisera

ISO 13485 EU MDR Documentation Toolkit Advisera

MDR Technical File and Documentation Template

Technical Documentation Template For Software Development prntbl

#regulatory #eumdr #template #technical #documentation #quality #

Requirements Checklist for Technical Documentation According to EU IVDR

MDR Article 120: Life After the Directive specculo

MDR Article 120: Life After the Directive specculo

How to build a Medical Device Technical Documentation (MDR 2017/745)

EU MDR FAQs: Industry Expert Insights Jama Software

We help you under EU Technical Documentation

We help you under EU Technical Documentation

We help you under EU Technical Documentation

We help you under EU Technical Documentation
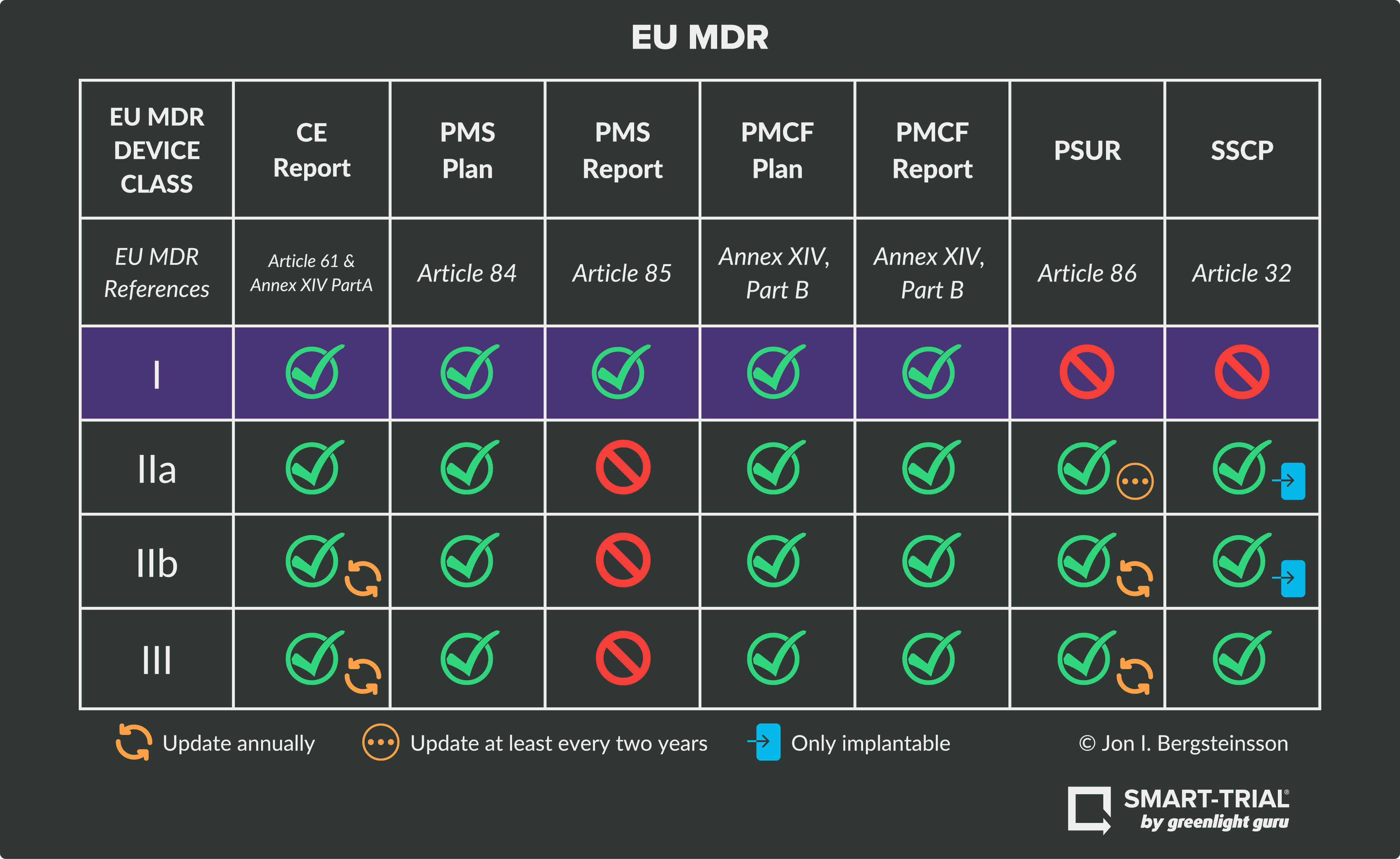
Ultimate Guide to Device Class Requirements under EU MDR

#eumdr #tdf #regulatoryaffairs #regulatory #technicaldocumentation #

#eumdr #medicaldevice #medicaldevices #submission #technical #

EU MDR Technical Documentation Outline specculo

EU MDR Technical Documentation Outline specculo

How to build a Medical Device Technical Documentation (MDR 2017/745)

How to build a Medical Device Technical Documentation (MDR 2017/745)

Comment créer une documentation technique sur les dispositifs médicaux

Complete Guide: Medical Device Classification EU MDR (Free PDF)

Medical Device Design History File Template prntbl

Mastering risk management for EU MDR: Technical documentation GSPR

GSPR General Safety And Performance Requirements EU MDR IVDR